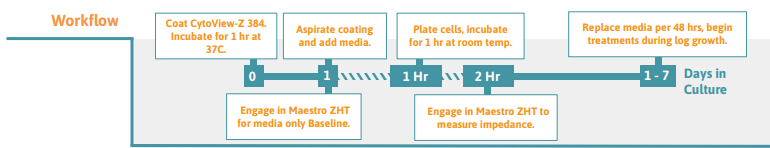
Preparing the CytoView-Z 384 Plate
- Pre-coat (50 µl per well is recommended) the entire well surface of the CytoView-Z 384 plate using fibronectin (or relevant surface coating for your cell type) to anchor adherent cells to the impedance plate surface. Tip: Reconstitute the relevant surface coating for the cell line in accordance to supplier recommendations.
- Centrifuge the plate at 300 xg for 2-5 minutes.
- Incubate the plate for 1 hour at 37°C.
- Pipette to remove excess surface coating solution from the plate.
- Add 50 µL of complete medium to the plate
- Centrifuge the plate at 300 xg for 2-5 minutes.
- Add 8 mL of sterile water to the on-plate reservoirs (3mL on each side, 1 mL on top and bottom) to increase humidity.
- Dock the plate in the Maestro ZHT to measure the media only (MO) Baseline. Transfer the plate to a biosafety cabinet when the Baseline recording is complete.
Plating Adherent Cells on CytoView-Z 384 Plate
- Thaw and culture the cells of interest in accordance with supplier recommendations, passaging as needed.
- Remove flasks of cultured cells from the incubator, aspirate the media and rinse with warmed PBS. Using trypsin, or another cell dissociating agent, detach and collect the cells from the flasks as per reagent recommendations.
- Remove a sample of the cell suspension and count the cells using a hemocytometer to determine both the viability and total number of viable cells. Tip: Ensure the cells are evenly suspended before removing an aliquot to count.
- Transfer the cell suspension to a 15 ml conical tube and centrifuge the cell suspension to pellet.
- Aspirate the supernatant, being careful not to disturb the cell pellet.
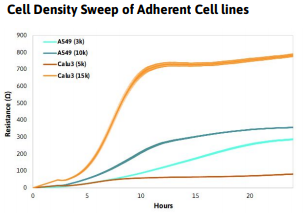
- Dilute the cell suspension in complete medium to a working concentration of cells per 25 µl. The working concentration should be the number of cells per well necessary to achieve 100% confluence within 24 hours, however this may vary based on specific experimental goals. Note: It is recommended to run a cell density sweep with a cell type first to determine the optimal number of cells for the working concentration and to inform future experiments. Tip: Use approximately 30- 40% of the amount of cells normally plated per well in a CytoView- Z 96-well plate
- Undock the CytoView-Z 384 plate from the Maestro ZHT once the media only baseline has been collected, and transfer the plate to a biosafety cabinet.
- Transfer the cell suspension to a trough for easy access by a multichannel pipette. Alternatively, divide the cell suspension evenly into microcentrifuge tubes that can be used with a multichannel pipette, providing enough volume to seed the cells with a 25 µL addition per well. Tip: Be sure to mix the cell suspension thoroughly before any addition to ensure even distribution of the cells. Dispense the cells directly in the middle of the well. Do not centrifuge plate after cell addition.
- After seeding cells on the plate, leave it to rest in the biosafety cabinet for 1 hour at room temperature. Tip: High well-number microtiter plates are sensitive to thermal gradients, which can cause edge effects.
- Dock the plate and impedance measurements will begin automatically upon plate engagement.
- For optimal cell health, 50% of the media should be changed every 48 hours.
Additional Notes
- Total Well Volume: The absolute minimum is 30 µL, as less has shown decreased growth rate. Between 50 µL and 130 µL per well is recommended. Maximum well volume is 150 µL.
- Edge Effects: For sensitive cell types, it is recommended to avoid edge wells, or to capture endpoint data, maintain the plate in incubator and remove only for periodic recordings.
Recommended densities for common cancer cell lines:
- A549 - 10,000 cells per well
- HeLa - 5,000 cells per well
- Calu3 - 15,000 cells per well

Required materials
Consumables
| Item | Vendor |
|---|---|
| CytoView-Z Plate | Axion BioSystems |
| Cell Culture Media | Various |
| 15 mL and 50 mL Centrifuge Tubes | Various |
| 1 mL Pipette Tips | Various |
Equipment
| Item | Vendor |
|---|---|
| Maestro Z | Axion BioSystems |
| AxIS Z | Axion BioSystems |
| Microscope | Various |
| 1 mL Micropipettor | Various |
| 8- or 12-channel Multiwell pipettor | Various |



